 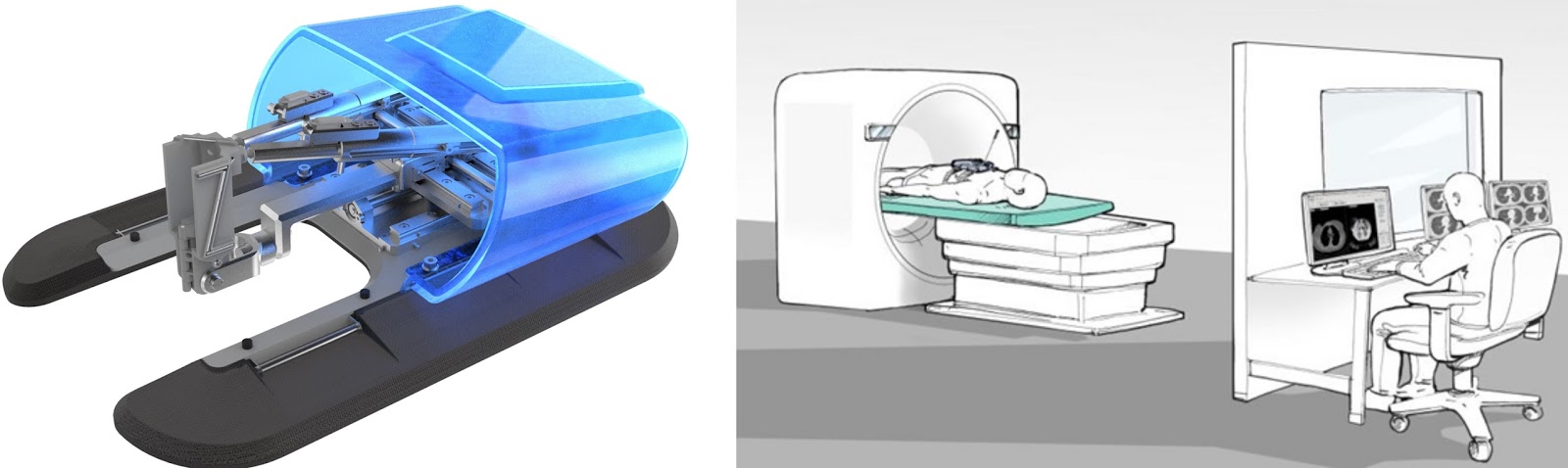
The Xact platform compensates for breathing and patient movement during the procedure for better accuracy and safety, Xact said. Once approved by the operator, the user can attach the appropriate tool to the robot to allow it to perform needle insertion and steering according to the plan, the company said. The system allows the physician to plan the procedure by selecting a target and entry point, after which the platform recommends a trajectory and verification checkpoints along the trajectory. The Caesarea, Israel-based company said its Xact robotics system was cleared for use in planning and accessing targets in the abdomen during computed-tomography-guided percutaneous procedures. unstable hypertension, unstable cardiac disease, etc.Xact Robotics said today it won CE Mark approval in the European Union for its robotics navigation and steering system designed for image-guided percutaneous procedures, and said it raised $5 million in a Series C round of financing. Subjects with an unstable medical condition, e.g.Subjects with a preexisting conditions, which, in the opinion of the investigator, may interfere with the conduct of the study.Subject with lesions in the central and peripheral nervous system and the spine.Subjects in whom the target is written 1 cm of a major blood vessel or major nerve.Subject is capable and willing to adhere to the study procedures.Subject is capable and willing to provide informed consent.Subjects undergoing CT-guided, minimally invasive percutaneous procedures in the interventional radiology suite, e.g., core biopsy, fine needle aspiration (FNA), tumor ablation, etc.The distance from the tip of the needle/tool to the target, once the XACT robot reaches the pre-defined target will be measured in order to determine system accuracy in quantitative terms using the system tools. The total time of the procedure will be recorded, as will the number of CT scans performed and the radiation dose (DLP and CTDI). This information will be used to calculate success rate.Īdditionally, the usability of the XACT device will be evaluated using a rating scale by assessing the ease of device setup, device operation, pre-operative planning, robot positioning, guiding and needle advancement. The investigator will review the final instrument position on the post-placement CT images to determine if the pre-operative planned target was reached. Investigators will screen patients based on the inclusion/exclusion criteria described below and the subjects' demographic, general medical history, medical condition/indication, coagulation factors, concomitant medications and vital signs will be obtained.Ĭlinical accuracy will be the primary efficacy endpoint and is defined as the ability to place the instrument or procedural tool at a location suitable for the planned intervention. 
The intention is to recruit subjects, which will cover a variety of CT-guided interventional procedures and a variety of different procedural tools that may be used with the device. Subjects undergoing CT-guided, minimally invasive percutaneous procedures in the interventional radiology suite, e.g., core biopsy, fine needle aspiration (FNA), tumor ablation, etc., and willing to sign an informed consent document will be screened for compliance with the study inclusion and exclusion criteria.Ī total of thirty (85) subjects will be enrolled in the study at 5 medical centers. The study will be approved by the Institutional Review Board (IRB) at each of the participating centers prior to patient enrollment. This is a prospective, single-arm study is to evaluate the safety, effectiveness and usability of the XACT device. 
Why Should I Register and Submit Results?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
